
SOTYKTU® (deucravacitinib) is indicated for the treatment of moderate-to-severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
Limitations of Use: SOTYKTU is not recommended for use in combination with other potent immunosuppressants.
Both studies assessed the responses at Week 16 compared with placebo for the two co-primary endpoints:
- The proportion of patients who achieved at least a 75% improvement in PASI scores from baseline (PASI 75)
- The proportion of patients who achieved an sPGA score of 0 (clear) or 1 (almost clear)
There were multiple ranked secondary endpoints, including:
- The proportion of patients who achieved PASI 75 at Week 16 and Week 24 vs apremilast
- The proportion of patients who achieved at least a 90% improvement in PASI scores from baseline (PASI 90) at Week 16 and Week 24 vs apremilast
- The proportion of patients who achieved a scalp-specific PGA (ss-PGA) score of 0 (clear) or 1 (almost clear) at Week 16 vs placebo and vs apremilast
Co-primary endpoints:
- PASI 75 at Week 16 for SOTYKTU vs placebo: PSO-1: 58% (n=193/330) vs 13% (n=21/166), P<0.0001; PSO-2: 53% (n=271/511) vs 9% (n=24/255), P<0.0001
- sPGA 0/1 at Week 16 for SOTYKTU vs placebo: PSO-1: 54% (n=178/330) vs 7% (n=12/166), P<0.0001; PSO-2: 50% (n=253/511) vs 9% (n=22/255), P<0.0001
Select secondary endpoints:
- PASI 75 at Week 16 for SOTYKTU vs apremilast: PSO-1: 58% (n=193/330) vs 35% (n=59/168), P<0.0001; PSO-2: 53% (n=271/511) vs 40% (n=101/254), P=0.0004
- PASI 90 at Week 24 for SOTYKTU vs apremilast: PSO-1: 42% (n=140/330) vs 22% (n=37/168), P<0.0001; PSO-2: 32% (n=164/511) vs 20% (n=50/254), P=0.0002
- ss-PGA 0/1 at Week 16 for SOTYKTU vs apremilast in patients with baseline ss-PGA score of ≥3: PSO-1: 70% (n=147/209) vs 39% (n=43/110) P<0.0001; PSO-2: 60% (n=182/305) vs 37% (n=61/166), P<0.0001
Statistical significance was not met for the following key secondary endpoints:
- PGA-F 0/1 (PGA-F score of clear or minimal disease) vs placebo (BL ≥3) at Week 16
- PSSD symptom score of 0 vs apremilast (BL ≥1) at Week 16
- POETYK PSO-LTE is an ongoing, open-label, long-term extension trial that enrolled participants from the Phase 3 POETYK PSO-1 and PSO-2 trials. All patients in PSO-1 and PSO-2 were eligible to enter the LTE trial after 52 weeks of treatment, regardless of initial treatment. At the start of the LTE (Week 52), 1221 patients were blindly switched from SOTYKTU, apremilast, or placebo to open-label SOTYKTU 6 mg once daily. Primary endpoints are incidence of adverse events and serious adverse events. Key secondary endpoints are sPGA 0/1 and PASI 75.
Data is presented as an interim analysis with a data cutoff of November 1, 2023. Outcomes were analyzed descriptively.
Week 16 Safety Profile1
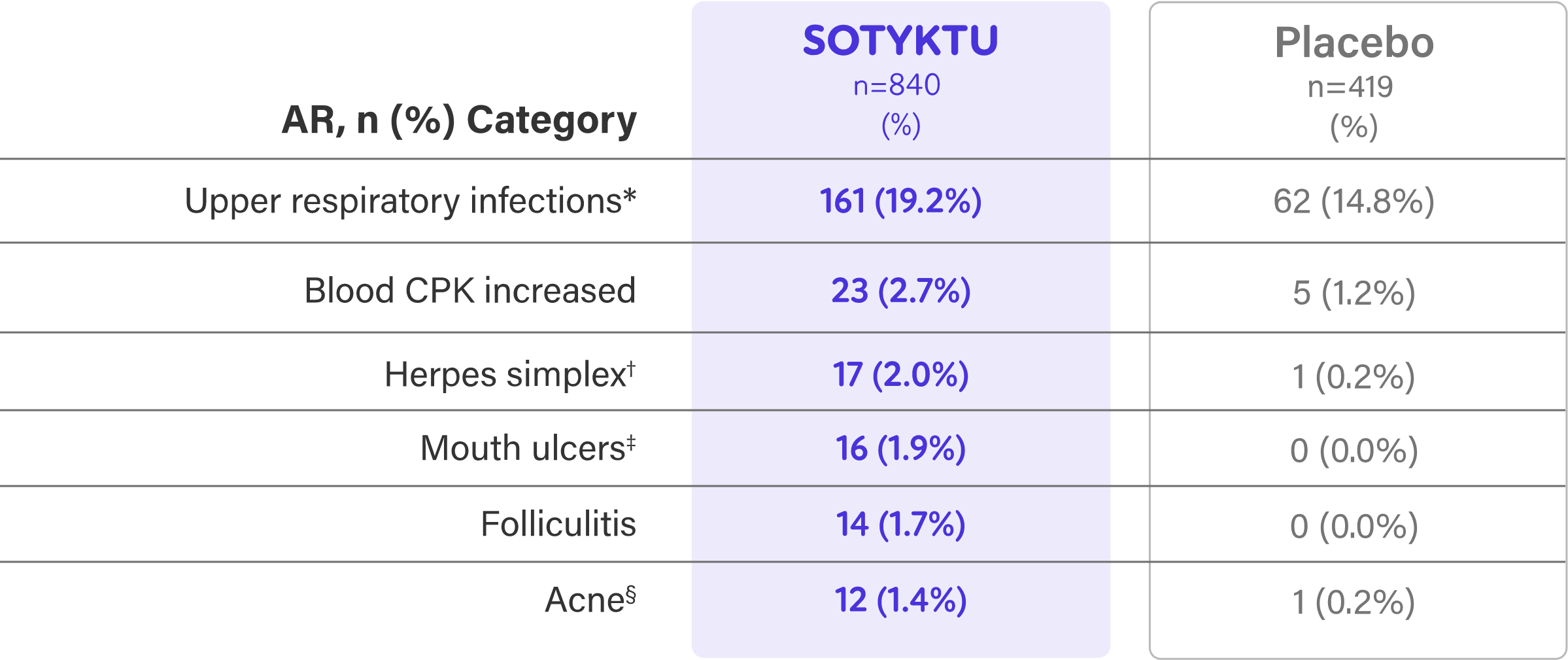
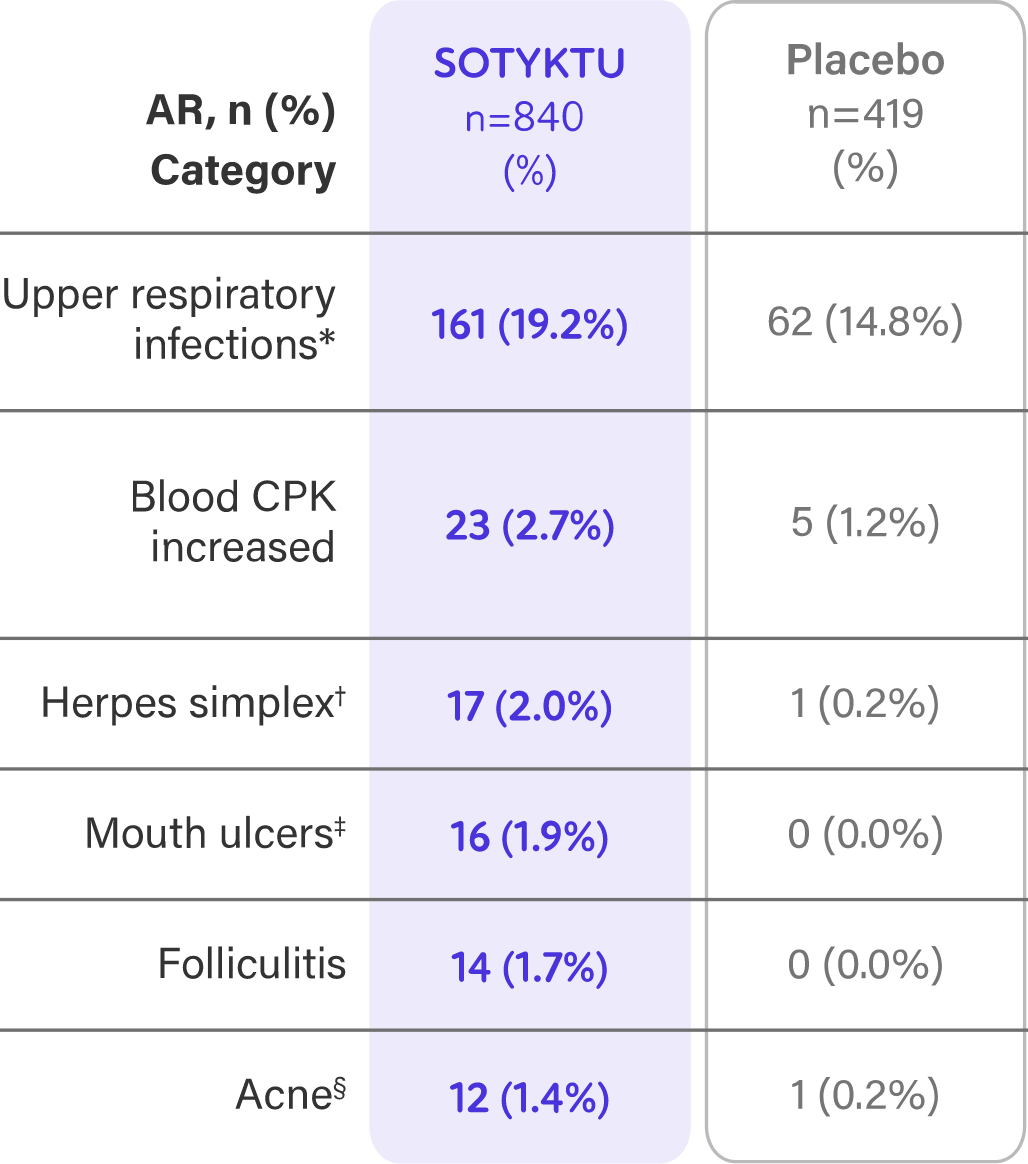
- Adverse reactions that occurred in <1% of patients in the SOTYKTU group were herpes zoster1
- Infections: In the first 16 weeks, infections occurred in 29% of the SOTYKTU group (116/100 PY) compared to 22% of the placebo group (83.7/100 PY). The majority of infections were non-serious and mild to moderate in severity and did not lead to discontinuation of SOTYKTU. The incidence of serious infections were reported in 5 patients (2.0/100 PY) treated with SOTYKTU, and 2 patients (1.6/100 PY) treated with placebo1
- Malignancies (excluding non-melanoma skin cancer) through Week 52 (total exposure of 986 PY with SOTYKTU) were reported in 3 patients treated with SOTYKTU (0.3/100 PY)1
- During clinical trials, including an open-label extension trial, 3 SOTYKTU patients (0.1/100 PY) developed lymphoma1
PY=patient year.
SOTYKTU is associated with the following Warnings and Precautions: Hypersensitivity, Infections, Tuberculosis, Malignancy including Lymphomas, Rhabdomyolysis and Elevated CPK, Laboratory Abnormalities, Immunizations, and Potential Risks Related to JAK Inhibition.
Please see additional Important Safety Information below.

Joe Gorelick, MSN, FNP-C
Compensated for his time
SPOTLIGHT CONTENT
Safety data, including from LTE
SOTYKTU™ Educational Series
for Nurse Practitioners and Physician Associates
Resources
for You and Your Patients
| IMPORTANT SAFETY INFORMATION and indication |
| IMPORTANT SAFETY INFORMATION |
| CONTRAINDICATIONS | ||||||||
| SOTYKTU is contraindicated in patients with a history of hypersensitivity reaction to deucravacitinib or to any of the excipients in SOTYKTU. | ||||||||
| WARNINGS AND PRECAUTIONS | ||||||||
| Hypersensitivity: Hypersensitivity reactions such as angioedema have been reported. If a clinically significant hypersensitivity reaction occurs, institute appropriate therapy and discontinue SOTYKTU. | ||||||||
| Infections: SOTYKTU may increase the risk of infections. Serious infections have been reported in patients with psoriasis who received SOTYKTU. The most common serious infections reported with SOTYKTU included pneumonia and COVID-19. Avoid use of SOTYKTU in patients with an active or serious infection. Consider the risks and benefits of treatment prior to initiating SOTYKTU in patients: | ||||||||
|
||||||||
| Closely monitor patients for the development of signs and symptoms of infection during and after treatment. A patient who develops a new infection during treatment should undergo prompt and complete diagnostic testing, have appropriate antimicrobial therapy initiated and be closely monitored. Interrupt SOTYKTU if a patient develops a serious infection. Do not resume SOTYKTU until the infection resolves or is adequately treated. | ||||||||
| Viral Reactivation | ||||||||
| Herpes virus reactivation (e.g., herpes zoster, herpes simplex) was reported in clinical trials with SOTYKTU. Through Week 16, herpes simplex infections were reported in 17 patients (6.8 per 100 patient-years) treated with SOTYKTU, and 1 patient (0.8 per 100 patient-years) treated with placebo. Multidermatomal herpes zoster was reported in an immunocompetent patient. During PSO-1, PSO-2, and the open-label extension trial, the majority of patients who reported events of herpes zoster while receiving SOTYKTU were under 50 years of age. The impact of SOTYKTU on chronic viral hepatitis reactivation is unknown. Consider viral hepatitis screening and monitoring for reactivation in accordance with clinical guidelines before starting and during therapy with SOTYKTU. If signs of reactivation occur, consult a hepatitis specialist. SOTYKTU is not recommended for use in patients with active hepatitis B or hepatitis C. | ||||||||
| Tuberculosis (TB): In clinical trials, of 4 patients with latent TB who were treated with SOTYKTU and received appropriate TB prophylaxis, no patients developed active TB (during the mean follow-up of 34 weeks). One patient, who did not have latent TB, developed active TB after receiving 54 weeks of SOTYKTU. Evaluate patients for latent and active TB infection prior to initiating treatment with SOTYKTU. Do not administer SOTYKTU to patients with active TB. Initiate treatment of latent TB prior to administering SOTYKTU. Consider anti‑TB therapy prior to initiation of SOTYKTU in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during treatment. | ||||||||
| Malignancy including Lymphomas: Malignancies, including lymphomas, were observed in clinical trials with SOTYKTU. Consider the benefits and risks for the individual patient prior to initiating or continuing therapy with SOTYKTU, particularly in patients with a known malignancy (other than a successfully treated non-melanoma skin cancer) and patients who develop a malignancy when on treatment with SOTYKTU. | ||||||||
| Rhabdomyolysis and Elevated CPK: Treatment with SOTYKTU was associated with an increased incidence of asymptomatic creatine phosphokinase (CPK) elevation and rhabdomyolysis compared to placebo. Discontinue SOTYKTU if markedly elevated CPK levels occur or myopathy is diagnosed or suspected. Instruct patients to promptly report unexplained muscle pain, tenderness or weakness, particularly if accompanied by malaise or fever. | ||||||||
| Laboratory Abnormalities: Treatment with SOTYKTU was associated with increases in triglyceride levels. Periodically evaluate serum triglycerides according to clinical guidelines during treatment. SOTYKTU treatment was associated with an increase in the incidence of liver enzyme elevation compared to placebo. Evaluate liver enzymes at baseline and thereafter in patients with known or suspected liver disease according to routine management. If treatment-related increases in liver enzymes occur and drug-induced liver injury is suspected, interrupt SOTYKTU until a diagnosis of liver injury is excluded. | ||||||||
| Immunizations: Prior to initiating therapy with SOTYKTU, consider completion of all age-appropriate immunizations according to current immunization guidelines including prophylactic herpes zoster vaccination. Avoid use of live vaccines in patients treated with SOTYKTU. The response to live or non-live vaccines has not been evaluated. | ||||||||
| Potential Risks Related to JAK Inhibition: It is not known whether tyrosine kinase 2 (TYK2) inhibition may be associated with the observed or potential adverse reactions of Janus Kinase (JAK) inhibition. In a large, randomized, postmarketing safety trial of a JAK inhibitor in rheumatoid arthritis (RA), patients 50 years of age and older with at least one cardiovascular risk factor, higher rates of all-cause mortality, including sudden cardiovascular death, major adverse cardiovascular events, overall thrombosis, deep venous thrombosis, pulmonary embolism, and malignancies (excluding non-melanoma skin cancer) were observed in patients treated with the JAK inhibitor compared to those treated with TNF blockers. SOTYKTU is not approved for use in RA. | ||||||||
| ADVERSE REACTIONS | ||||||||
| Most common adverse reactions (≥1% of patients on SOTYKTU and more frequently than with placebo) include upper respiratory infections, blood creatine phosphokinase increased, herpes simplex, mouth ulcers, folliculitis and acne. | ||||||||
| SPECIFIC POPULATIONS | ||||||||
| Pregnancy: Available data from case reports on SOTYKTU use during pregnancy are insufficient to evaluate a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Report pregnancies to the Bristol-Myers Squibb Company's Adverse Event reporting line at 1-800-721-5072. | ||||||||
| Lactation: There are no data on the presence of SOTYKTU in human milk, the effects on the breastfed infant, or the effects on milk production. SOTYKTU is present in rat milk. When a drug is present in animal milk, it is likely that the drug will be present in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for SOTYKTU and any potential adverse effects on the breastfed infant from SOTYKTU or from the underlying maternal condition. | ||||||||
| Hepatic Impairment: SOTYKTU is not recommended for use in patients with severe hepatic impairment. | ||||||||
| SOTYKTU is available in 6 mg tablets. | ||||||||
| INDICATION | ||||||||
| SOTYKTU® (deucravacitinib) is indicated for the treatment of moderate-to-severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. | ||||||||
| Limitations of Use: | ||||||||
| SOTYKTU is not recommended for use in combination with other potent immunosuppressants. | ||||||||
| Please click for U.S. Full Prescribing Information, including Medication Guide, for SOTYKTU. | ||||||||
References:
|
SOTYKTU, SOTYKTU 360 SUPPORT, and related logos are trademarks of Bristol-Myers Squibb Company.
All other trademarks are property of their respective owners.
© 2024 Bristol-Myers Squibb Company.
1787-US-2400146 04/24

